FDA Approves New Obesity Pill: A Medical Breakthrough
With the FDA's recent approval of Foundayo, a new obesity pill, patients gain a promising alternative to injectable treatments. This development could revolutionize obesity management, offering easier access and potentially improving patient adherence.
Introduction
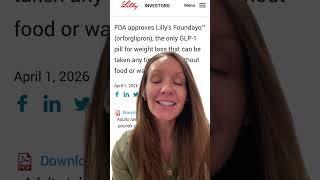
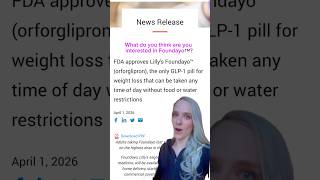
The FDA has approved a new obesity pill, Foundayo, offering patients a promising alternative to injectable treatments. This development is significant as it potentially transforms obesity management by providing easier access and potentially improving patient adherence.
Background/Context
Obesity is a chronic condition affecting over 40% of adults in the United States, according to the Centers for Disease Control and Prevention (CDC). Traditionally, treatment options have included lifestyle changes, medications, and surgery. Injectable drugs like Wegovy have been at the forefront due to their effectiveness, but they present challenges related to administration and accessibility.
Historically, the development of oral obesity medications has been fraught with challenges, including side effects and limited efficacy. Previous oral medications have faced scrutiny, and some were withdrawn from the market due to safety concerns. The approval of Foundayo marks a new era in obesity treatment, representing a significant leap forward in pharmaceutical advancements.
Current Situation
Foundayo's approval by the FDA was expedited through the fast-track process, highlighting the urgency of addressing obesity. According to industry sources, Foundayo offers comparable efficacy to the injectable Wegovy, with clinical trials indicating significant weight loss among participants. The pill is expected to hit the market soon, providing a convenient alternative for patients who struggle with injections.
Pharmaceutical companies are gearing up for the commercial launch, with widespread anticipation from healthcare providers and patients alike. The competition between Foundayo and other treatments, such as Wegovy, is expected to drive innovation and potentially reduce costs for patients.
Deep Analysis
The underlying mechanisms of Foundayo involve targeting specific hormonal pathways that regulate appetite and energy balance. According to research findings, the pill modulates the action of hormones like GLP-1, which plays a crucial role in satiety and glucose metabolism. This approach aligns with the current understanding of obesity as a complex interplay of genetic, hormonal, and environmental factors.
Experts from the American Medical Association suggest that Foundayo's oral administration could improve patient adherence, as it eliminates the need for injections. However, they also caution about the importance of monitoring for potential side effects, as with any new medication.
Impact/Outlook
The short-term impact of Foundayo's approval is expected to be significant, with a potential shift in the treatment landscape. Healthcare providers are optimistic about the increased options available to patients, which could lead to better management of obesity-related health conditions such as diabetes and cardiovascular diseases.
In the long term, the success of Foundayo could pave the way for further research into oral treatments for obesity and other metabolic disorders. The pharmaceutical industry is likely to invest more in developing similar medications, potentially broadening the arsenal of tools available to combat obesity.
Practical Implications
For healthcare providers, the introduction of Foundayo means re-evaluating treatment protocols and educating patients on the new option. Patients are advised to consult with their healthcare providers to determine if Foundayo is suitable for their individual needs. Lifestyle modifications, including diet and exercise, remain integral components of effective obesity management.
Policymakers and insurance companies may need to consider the implications of coverage for new obesity treatments, balancing cost with potential health benefits.
Key Takeaways
- Foundayo's FDA approval represents a breakthrough in oral obesity treatment.
- The pill offers an alternative to injectable treatments like Wegovy, with comparable efficacy.
- Oral administration may improve patient adherence but requires monitoring for side effects.
- The approval signals a potential shift in the obesity treatment landscape, encouraging further research and development.
- Healthcare providers and patients must collaborate to integrate new treatments with lifestyle modifications.